VERBAL FLUENCY, CLUSTERING AND SWITCHING IN PERSONS WITH DEPRESSION AS INDICATORS FOR COGNITIVE IMPAIRMENTS
Abstract
Introducere:. Fluența verbală (FV) reprezintă capacitatea unei persoane de a spune cuvinte pe baza unor criterii date. FV fonematică presupune oferirea de cuvinte care încep cu o literă specifică – de obicei FAS, iar fluența verbală semantică se referă la o categorie – ex. animale, fructe. Performanța la fluența verbală este sensibilă în cazul tulburărilor depresive datorită relației dintre fluență și disfuncțiile din depresie pe palierul atenției susținute, concentrării, vitezei de procesare și memoriei. Sarcinile de fluență verbală (FV) sunt des folosite pentru evaluarea funcționării cognitive și executive în diferite tulburări psihiatrice. Obiective: Acest studiu ilustrează fluența verbală, atât componenta fonematică, cât și cea semantică pentru un eșantion de persoane cu tulburări depresive din populația românească prin analiza cantitativă și calitativă, drept indicatori ai disfuncțiilor cognitive și a deficitelor executive. Material și metodă: Studiul presupune analiza producțiilor de fluență verbală pentru 41 de persoane fără istoric de boli psihice sau de tulburări de dispoziție și 32 depresivi. Criteriile de includere pentru depresie au fost diagnosticul clinic pe baza criteriilor ICD-10, scala DASS-21R și MADRS. Criteriile de excludere au fost vârsta (sub 18 ani – peste 65 de ani) și existența altor tulburări psihiatrice. Ambele grupe au fost evaluate cu scala de fluență verbală. Au existat în total 6 administrări pentru fluența verbală, varianta literelor FAS, respectiv a literelor CTL. Pentru fluența verbală semantică a fost utilizată categoria fructe. Metoda de cotare urmează modelul clasic propus de Benton dar și clusterii și schimbările din modelele lui Abwender(2001) și Troyer(1997). De asemenea memoria verbală a fost testată prin California Verbal Learning Test (CVLT) din cadrul Bateriei CogTest. Rezultate: Grupul cu depresie a obținut scoruri mai mici față de grupul participanților din lotul de control. Numărul de clusteri și de schimbări folosite sunt mai reduse în grupul persoanelor cu depresie, și descrește o dată cu severitatea simptomelor. Performanța la fluența verbală nu a fost influențată de nivelul de educație a participanților. Concluzii: Eșantionul persoanelor cu depresie din populația românească prezintă disfuncții cognitive reflectate prin scorul total obținut la testul de fluență verbală. Aceste rezultate sunt în concordanță cu cele din literatură și confirmă validitatea sistemului de cotare pe bază de clusteri și schimbări drept indicatori ai disfuncției cognitive.
Acknowledgements:
This paper was published under the frame of European Social Fund, Human Resources Development Operational Programme 2007-
2013, project no. POSDRU/159/1.5/S/ 138776.
INTRODUCTION
Neuropsychological dysfunction is considered a central deficit in major mood disorders. Depression is associated with neurocognitive deficits. Studies on depression suggest deficits in executive functioning, deficits in the area of memory, psychomotor functioning, generalized intellectual impairments, deficits in complex problem solving, slowing thinking, compromised ability to think, disturbed sustained attention, affective process and impaired decision-making(1). The verbal fluency task might help in setting a differential diagnosis between dementia and depression (2, 3). As previously observed some cognitive deficits still persist after remission of depression (4, 5, 6). Verbal fluency, reaction time, impaired recognition of abstract visual information from long-term memory are few of them(4).
Concerning the functional and structural substrates of the brain in depression, studies using PET (Positron Emission Tomography) and fMRI (Functional Magnetic Resonance Imaging) provide evidence of cortical and subcortical dysfunction: orbitofrontal cortex, medial thalamus, anterior and posterior cingulated cortices, ventrum striatum (6, 7).
Models of verbal fluency performance were developed from observation of performance variables in different population. Verbal fluency represents the ability of a person to express a set of words based on requirements established by an evaluator.
One of the most commonly used measures of phonemic verbal fluency is Controlled Oral Word Association Test, also referred as COWAT (8). It has been used since the beginning as an aid in determining neurocognitive ability. Verbal fluency task (VFT) or COWAT is wildly used in order to assess cognitive and executive functioning, deficits in different populations with mental health illness (9, 10, 11, 12, 13, 14) and others impairments (15, 16, 17, 18, 19, 20, 21, 22). Due to its easy application verbal fluency task has been extensively used in clinical settings and also in research study designs.
The purpose of this task is to evaluate spontaneous production of words within a specific period o time, usually one minute (19). The two forms of the fluency task most commonly employed are semantic and phonemic fluency.
Performance on verbal VFT could be in particular sensitive to depressive disorder due to the relation between fluency and deficits in depression: sustained attention, concentration, retrieval and speed (23). Verbal fluency measurements are considered to impose comparable demands upon executive and supervisory processes because both requires efficient organization of verbal retrievals and recall, self monitoring aspects of cognition, effortful self-initiation and inhibition of response when appropriate(24). Studies done on patients with Alzheimer’s disease (3, 10, 25, 26) showed that they rarely generate words from semantic categories. They simply cannot answer semantic probe questions, implying that fluency performance is dependent on semantic memory stores. This observation confirmed the role of cognitive processes in verbal fluency. Thus, in case of semantic verbal fluency few requirements need to be fulfilled (27): an intact semantic store for supplying a knowledge base of related words and an effective search process to access and retrieve this information. Semantic fluency requires “searching for semantic extension of a target super ordinate, depending intrinsically upon the integrity of semantic association within the lexicon” (24). So the incapacity to generate words in the verbal fluency task is determined either by deterioration of the stored knowledge base, or by inefficient search. Also, both causes might coexist. In the case of semantic fluency, the main cause is relatively more dependent upon the integrity of semantic memory. In the case of phonemic verbal fluency, executive dysfunctions are present. Frontal structures, as structural components of the brain, play an important role in the executive function. As Henry (24) underlined a pattern of comparable impairments upon tests of phonemic and semantic fluency of patients with depression may reflect executive dysfunction. In terms of brain components, semantic fluency was more impaired after focal temporal damage, and phonemic verbal fluency in the case of frontal lobes damages.
The verbal fluency task sought to reflect the integrity of brain circuits and cognitive and executive functioning. But reported norms might be tricky and quantitative scores are insufficient.
Improved and complex ways of scoring VF are available. Qualitative interpretation of VF is more accurate (28). Clustering and switching are more sensitive to deficits in the verbal fluency task and consequently better predictors for cognitive impairments in depression. Clustering operates the previous store concept (27), while switching covers the search process. Troyer (29) define clustering as the production of words within semantic or
phonemic subcategories and switching as the ability to shift efficiently to a new subcategory. In Troyer’s (30) opinion switching scores assesses executive function such as „ability to disengage from a previous strategy and the cognitive flexibility to select a new word retrieval strategy quickly and efficiently” (31). Abwender et al. (32) expanded the scoring system by including additional instances of clustering, more exact task –discrepant clustering and task consistent clustering.
Although studies on verbal fluency implies an easy study design and normative data were collected from different population such as Polish (33), Japanese (34), Danish (35), Mexican (36), Portuguese (37), German (38) etc., poor data are available for the Romanian population.
Few could be mentioned for cognitive impairments in elderly (39), schizophrenia patients (40), or partial use of VFT as a sequence in another scale (41). Based on our knowledge, there is no investigation for verbal fluency in persons with depression, in Romanian population.
The aim of this study is to illustrate verbal fluency in a Romanian sample of persons with depression using both COWAT quantitative scores and Troyer’s (30) and Abwender’s (32) qualitative scores as possible indicators for cognitive impairments.
MATERIALAND METHODS
Participants
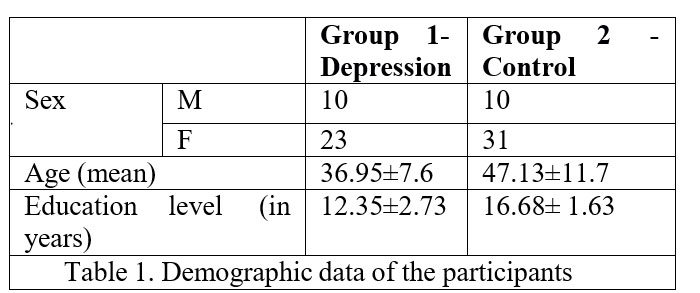
There were two groups of participants. The control group has 41 persons with no history of personal or family mental health illness. The second group was represented by psychiatric inpatients from Cluj County Emergency Hospital diagnosed with depression. The second group has 32 participants (Table no.1).
Procedure
Each participant was individually administered a brief battery of tests. This battery includes: two scales for depression – the Romanian version of Depression Anxiety
Stress Scale – DASS-21 R (42), and the Romanian version of SIGMA/MADRS – Montgomery & Asberg Depression Rating Scale 10 (43), a Word Memory test– California Verbal Learning Test, the computerized form from CogTest – and the verbal fluency task, both phonemic and semantic. Phonemic verbal fluency task (PVF) consists of three different letter tests which requires participants to tell as many words as possible, beginning with a specific letter (usually C-F-L) in a specific amount of time – 60 seconds for each trial (31). Alternate forms are available using letters P-R-W or F-A-S. Before the task is applied, short instructions are given. Participants are prohibited from saying proper nouns or words that differs only by ending – call, called; repetitions are not allowed(19).
Semantic verbal fluency (SVF) is constrained by a category, for example animals, fruits, vegetables etc. The instructions are the same.
In our study for phonemic verbal fluency task there were two different sets– one contained FAS letters and the others one CTL letters. This decision was based on the data from literature(31) and on the characteristic of the frequency of the letters in Romanian languages. Although the literature also suggests PRW letters for testing PVF, words starting with “W” are almost inexistent in Romanian language. As long as FAS and CFL are the others most used letters reported in the COWAT research reports, we considered it a better choice. But we also added the letter “T” for testing in order to use letters from the end of the alphabet. This technique allows comparisons between results from different sets of admission. FAS and PRW versions of COWAT had been previously compared (31), however FAS and CFL or CTL have not been directly compared for qualitative score equivalence. For SVF the category was fruits. Each participant was recorded during VFT admissions. We offered the participants 1 minute for each letter / category.
The performance in verbal fluency is calculated by the total number of words produced across the three trials (letters). Any unacceptable responses such as repetition – persistence, mismatched words, rule violations are drop from the total score (8). We scored the VF for total numbers of words in each admission – FAS/CTL version as the COWAT general procedure.
Additionally we used clusters and switching scores based on Troyer et al (30), respectively Abwender et al (32) models. For COWAT we counted words as cristal – cristalin, lampă – lampadar for PVF and in case of SVF the name of the tree was associated with the name of the fruits – ex. afin, zmeur, măr, păr. For PVF we did not count words considered a rule violation such us: same words with different ending, acelaşi – aceeaşi, alint – alintat, lingură – linguriţă, grammatically incorrect words – totdeauana – instead of întotdeauna, proper nouns – Ana, Făget, Fântânele, repetition –acasă – acasă, alint- alint. We scored word considered regionalism or with forms used by different groups – topogan –tobogan, lele. We also scored words from other languages but wildly used in conversation such as tomate, full, fake.
According with Troyer` system of scoring – “phonemic clusters are defined as groups of two or more successively generated words that (1) begin with the same first two letters (e.g., Church, change, charge); (2) differ only by their vowel sounds (e.g., fit, fat, foot); (3) rhyme (e.g., stand, sand); and (4) are homonyms (e.g., some, sum). These authors also introduced a cluster size score. Cluster size is determined by the number of words produced in each cluster beginning with the second word. For example the three-word cluster including car, carpet, and carrot would have a cluster size of two. Switching is defined as the ability to shift efficiently to a new category or subcategory, once a previous category is no longer productive. Switches are calculated as the number of transitions between clusters, between clusters and single words, and between single words” (31). In this study examples of clusters are: toc, lamă (homonyms), fată, facă, fadă (same two letters), car – cer, fix-fax(vowel difference), legat, legănat (rhyme).
Based on Abwender et al. (32) model additional instances of clustering were analyzed. This model proposed “additional criteria for phonemic cluster scores including (1) words that begin with the same sound, even if the spelling is different (e.g., school, skin); (2) words that end with the same sound (e.g., farther, future, feather); and (3) groups of words in which the second letter follows an alphabetical progression (e.g., about, across,
adore)” (31) Because Romanian is a phonetic language, with almost perfect phoneme-grapheme correspondence, first additional criteria proposed by Abwender is not so frequent, but few e.g are kiwi, xerograf, chenar. For the others e.g. are: amar, acar, avar (same ending), sardea, sborșit, scadent (alphabetical progression for second letter).
In his scoring system, Abweder distinguishes between task-discrepant clustering and task-consistent clustering in the analysis of clusters. Task-consistent clustering is a phonemic clustering on a phonemic task (e.g., COWAT) or semantic clustering on a semantic task (e.g., animal naming subcategory). By contrast, task- discrepant clustering refers to semantic clustering on the COWAT (e.g., potato, peanut) or phonemic clustering on a semantic fluency task (e.g., apple, apricot). For our study we score task-discrepant clustering in case of “caisă, căpşună” on C letter for COWAT, or “pară, pepene, piersică” – alphabetical order on SVF.
In order to collect accurate data, we used word count and alphabetical order from Office Word Program. Data were collected and Statistical Analysis was performed with SPSS. DASS -21R scoring was performed according with the Romanian scoring manual by two separate evaluators. MADRS was applied and scored by a clinician. DASS 21R and MADRS were compared for inclusion-exclusion criteria. In cases where the participant invited to take part in the study as a control recorded high scores at MADRS and at DASS- 21, they were distributed to the participants in the depression group.
Ethical Considerations
The study performs a non – interventional design. Methodology of the study and informed consent models were approved by Ethical Board of “Iuliu Haţieganu” University of Medicine and Pharmacy from Cluj-Napoca. This study guaranteed privacy rights of the volunteer participants and population with depression. Each participant signed an informed consent paper. There were neither any financial benefits nor other kind for the participants of this study.
RESULTS
The correlation between scores in MADRS and DASS
was performed. There is a high positive correlation r (73) = .85, p= 0.01.
Age rage was between 19 (minim) and 65 years old (maximum). The mean age for control group was 36.95(±7.6) years and for group with depression was 47.13(±11.7) years.
Quantitative analysis of verbal fluency
No statistical significant correlation was between the number of additional words in CVLT and PVF – FAS and CTL versions (Table no. 2). But a negative relation was observed. The CTL version of verbal fluency was -0.021, N=73.

Table 2. Correlation data between PVF- FAS version and word memory test – additional words
We found poor correlation between California Verbal Learning Test (CVFL – words memory test) and verbal fluency (phonemic and semantic). The Pearson coefficients are: r (73) = .40, p= 0.01. – PVF- CTL version and WLM Total Learning (over all trials), r (73) = .36, p=
0.01 – PVF – FAS version and WLM Total Learning (over all trials), r (73) = .48, p= 0.01– SVF and WLM Total Learning (over all trials). CVFL had no statistically significant correlation with Troyer’s cluster and switching system and none with Abwender’s task discrepant and task consistent system of clustering.
But high positive correlation was between the two versions of verbal fluency- FAS and CTL, N =73 (Table no 3).
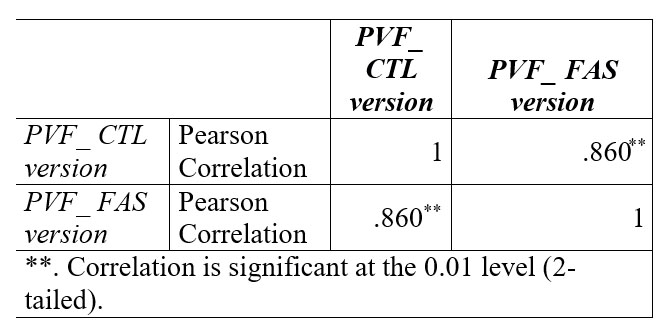
Table 3. Correlation data between FAS and CTL version of phonemic verbal fluency
The correlation between PVF (both FAS and CTL version) and SVF is also statistically significant but with smaller coefficient. Table 4 shows the correlation between PVF – FAS version and SVF. For PVF – CTL version and SVF correlation coefficient is r (73) = .61, p= 0.01.
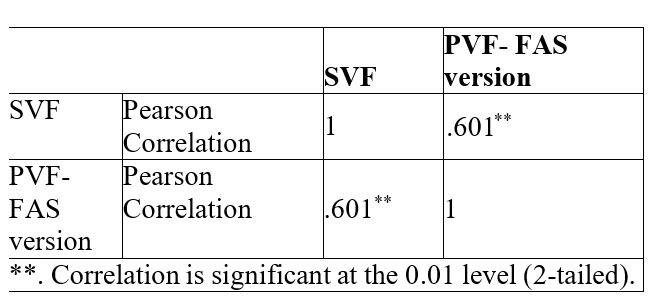
Table 4. Correlation data between phonemic and semantic verbal fluency
The group with depression (M=28.5, SD 12) performed worst in PVF – FAS version than the control one (M = 44.6, SD = 10.6), t(71) = – 6.084, p = .05.
The same results are for PVF – CTL version, group with depression (M = 29.09, SD = 11.5) performs worse than the control one (M = 44.90, SD = 11), t(71) = 5.94, p = .05.
Similar results were obtained in case of SVF (Table no. 5). The group with depression (M = 11.56, SD = 3.73) performs worse than the control one (M = 17.8, SD = 4.64).
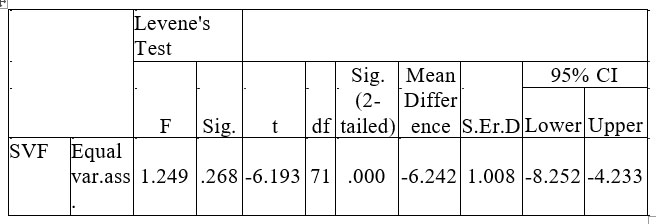
Table 5. Differences data between group of person with depression and the control for SVF
The number of words produced by the persons with depression is much smaller in phonemic verbal fluency, both in FAS and CTL versions. By contrast, rule violation, repetitions and proper nouns are more. Group with depression (M=1.28, SD 1.32) gave more FAS – repetitions, than the control one (M = .37, SD = .662), t(71) = 3.57, p = .05; on FAS – rule violation – group with depression (M=.72, SD 1.27), control group (M = .22, SD = .613), t(71) = 2.20 p = .05; on FAS – proper nouns – group with depression (M=1.81, SD 2.52), control group (M = .29, SD = .64), t(71) = 3.71 p = .05; on CTL – rule
violation – group with depression (M=.07, SD 1.24), control group (M = .15, SD = .52) ), t(71) = 3.09 p = .05; on CTL – repetitions – group with depression (M=1.0, SD 1.12), control group (M = .39, SD = .62) ), t(71) = 2.69 p = .05; on CTL – proper nouns – depression (M=1.41, SD
2.72) control (M = .22, SD = .65), t(71) = 2.69 p = .05.
Data are similar in the case of semantic verbal fluency – on SVF repetitions – group with depression (M=.84, SD 1.41), control group (M = .34, SD = .52), t(71)
= 2.02 p = .05; on SVF – rule violation – group with depression (M=.44, SD 1.64), control group (M = .02, SD = .15) ), t(71) = 2.69 p = .05. All results were statistically significant.
The results on PVF and SVF did not interfere with education level r (73) = – .09, p= 0.01
Qualitative analysis of verbal fluency
Correlations were calculated between the systems proposed by Troyer et al. (30), respectively Abwender et al. (32) and phonemic verbal fluency. Table no. 6 reflects the correlation between each version of verbal fluency, with each system. C4 also correlate with task-discrepant clustering
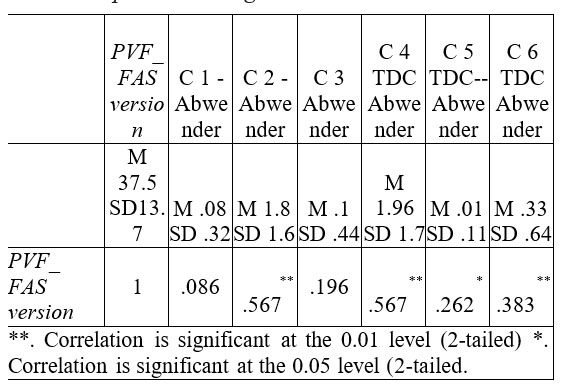
Table 6. Correlation between PVF- FAS version and
Abwender’s cluster system
Correlations between PVF – CTL version and Abwender’s clustering system are to be found in the Table no. 7.
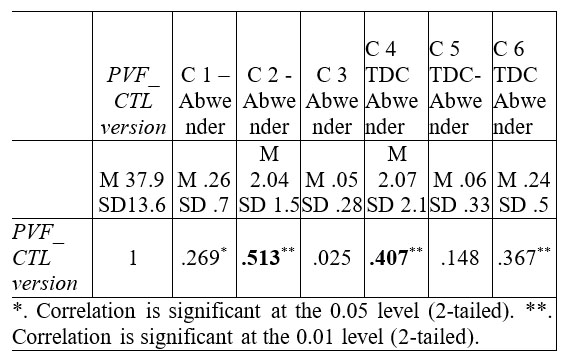
Table 7. Correlation data between PVF- CTL version and
Abwender (2002) VF scoring system
Correlations between Troyer system and PVF_ FAS
version are to be found in Table no. 8.

Table 8. Correlations data between PVF_ FAS version and cluster and Troyer’s clustering and switching system
We found similar results in case of CTL version (Table no.9).
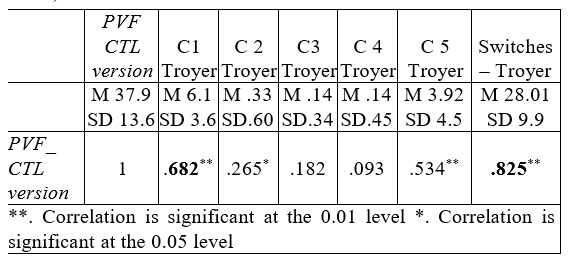
Table 9. Correlations between PVF_ CTL version and
Troyer’s clustering and switching system
Using the qualitative system proposed by Troyer et al. (30) we found statistically significant differences between the patients with depression and the control group on most of the clusters. Data are in detail presented bellow.
For phonemic verbal fluency FAS version and C1 (words beginning with the same first two letters) – group with depression (M=3.78, SD 2.79), control group (M = 6.9, SD = 3.4), t(71) = – 4.2 p = .05; C2 (differ only by their vowel sounds) – group with depression (M=.03, SD
1.7), control group (M = .6, SD = .68 ), t(71) = – 5 p = .05, C3(rhyme) – group with depression (M=.16, SD .44), control group (M = .22, SD = .52 ), t(71) = – .54 p = .05; C4(homonyms) – group with depression (M=.03, SD .17), control group (M = .73, SD = 3.7), t(71) = – 1.05 p = .05; C5 (number of words produced in each cluster beginning with the second word) – group with depression (M=1.94, SD 3.6), control group (M = 4.34, SD = 3.2), t(71) = – 2.9 p = .05; S (Switching) – group with depression (M=24.5, SD 10.8), control group (M = 24.4, SD = 11.03.), t(71) = – 3 p = .05.
For phonemic verbal fluency CTL – version and Troyer’ system results are similar.
Regarding the system proposed by Abwender et al. (32) we found statistically significant difference between the patients with depression and the control group on most of the cluster. Ross noted “there is evidence to suggest that each task is sensitive to different disease process and distinct neuroanatomical substrates” (44). The data are shown below:
For phonemic verbal fluency FAS – Version – C1 (words that begin with the same sound, even if the spelling is different) – group of person with depression (M=.6, SD .35) control group (M = .1, SD = .30), t(71) = – 4.5 p = .05;
C2 (words that end with the same sound) – group of person with depression (M=1.2, SD 1.2) control group (M = 2.2, SD = 2.8 ), t(71) = – 2.6 p = .05, C3 (groups of words in which the second letter follows an alphabetical progression) – group of person with depression (M=.0, SD
.0) control group (M = .17, SD = .58 ), t(71) = – 1.6 p = .05; task-discrepant clustering – same category – group of person with depression (M=1.2, SD 1.4) control group (M = 2.5, SD = 1.7), t(71) = – 3.5 p = .05; task-discrepant clustering – antonyms – group of person with depression (M=.0, SD 0.0) control group (M = .02, SD = .15), t(71) = – .8 p = .05; S task-discrepant clustering – category hierarchic – group of person with depression (M=.25, SD
.44) control group (M = .39, SD = .77), t(71) = – .9 p = .05. Similar results are in case of verbal fluency CTL version.
Using Abwender’s task consistent clustering and task discrepant clustering system on SVF we found no difference between group of person with depression (M=.56, SD .56) and control group (M = .56, SD = .67.), t(71) = – .01 p = .05 for task discriminating clustering and statistically significance with task consistent clustering – group of person with depression (M=2.6, SD 1.8) vs. control group (M = 4, SD = 1.4.), t(71) = – 3.5 p = .05
DISCUSSIONS
The descriptive data for the present study are similar with other results found in literature (30, 32, 44, 45, 46). The results on verbal fluency were not sensitive to the education level of the participants. The differences are similar for phonemic verbal fluency and semantic verbal fluency. In the case of our study, the number of males was equal for each group, but less than the number of women. The distribution is considered normal as long as depression is usually more common in women before the senior age and more common in elderly men. Numerous studies reported no effect of age (47)(48), the same was for our study. Consequently the age could not determine verbal fluency results. The mean age of group with depression (47.1) is under 50 and Rodriguez-Aranda (49) report a decline of this verbal ability especially after 60 years which accelerates along the late 80s.
No difference was recorded between PVF – FAS Versions and PVF – CTL Version. With a high correlation coefficients (r=.86, p=0.01) results on phonemic verbal fluency are similar in the clinical setting even though we choose to use letters FAS or letters CTL. This is in concordance with results from Abwender (32), who examined COWAT version CFL and PRW and generally found them to be equivalent with respect to qualitative and quantitative scores. In our study we found a correlation between phonemic verbal fluency and semantic verbal fluency. The coefficient r =.61 is statistically significant, though not as strong as the r coefficient obtain between FAS- CTL version. This suggests that in most of the cases the impairments are dependent on the neural circuits that are responsible for PVF and for SVF. This support Henry`s idea (24) which states that in the case of patients with depression, PVF and SVF may not reflect executive function, but more generalized impairments.
Contrary to others research reports (30, 32, 46) we found a poor correlation (r = .40, p= 0.01) between word memory test, respectively the total correct answers across trials 1-5 on CVLT and the total score on verbal fluency. The coefficient is similar for the two versions of PVF and higher for SVF. A possible explanation of this result might be the dimension of the groups. In this study the total number of participant is under 100 while others studies have larger number of participants. A larger study cohort could result in a higher correlation. A second explanation might be the characteristics of participants. Correlation between CVLT and verbal fluency were calculated only on healthy person while this study also implies persons with depression. Consistent with Ross we found no correlation between CVLT and the cluster and switches scores from Troyer`s and Abwender`s system. Ross (31) reported no correlation between total recalls on the CVLT and semantic clustering on COWAT. We consider appropriate to have a negative relation between the additional words from CVLT and the total score numbers for PVF. Additional words on CVLT’s list reflects the low ability to control and to inhibit distracters stimuli, while a proper control facilitates accuracy in word production in PVF.
As it was expected the overall results for total words score on PVF and SVF in group with depression are poorer than those in the control group. Verbal fluency deficits qualify as differential deficits relative to cognitive functioning. Consistent with other researches (23, 38, 47) both types of verbal fluency are impaired in depressed people. We found no difference between results on PVF and SVF in depressed people. Similar with Crowford`s data (24) depression might be associated with degraded semantic stores. Rules violation and usage of proper nouns are higher in persons with depression. This indicates either the incapacity to control interferences or the incapacity to keep the rule active in the short term memory. Despite the fact that those process are different in terms of brain structures and networks (34) both of them reflect cognitive impairments. As a measure of neurocogntive functioning, VF is often used to compare neurological impaired participants with normal controls. But sample sizes “have a propensity for distortion or bias due to demographic sample”(21), so qualitative scores are more relevant.
The difference is visible also in the case of clustering and switch scores employing the system developed by Troyer et al. (50) and clustering with TCC and TDC based on the system proposed by Abwender et al. (32). Qualitative analysis of VFT offers a detailed picture. It is important to mention the fact that in case of Abwender’s TDC cluster for groups of words in which the second letter follows an alphabetical progression have a score of 0 for depression (M=.0, SD .0). It is also the case for task-discrepant clustering – antonyms. Consistently with Abwender et al. (32) in the present study every person recorded at least 1 cluster for TCD or TDC in PVF.
For semantic verbal fluency extreme value were recorded. The highest number was for a non-depressive person – 29 words with no repetition and no rule violation. The lowest score was the case of depression with just 2 correct words, and 9 rule violations. The increased number of rule violation on category fluency could be an indicator for additional impairments. As Henry (24) states “semantic fluency is relatively more dependent upon integrity of semantic memory”. So in the case of patients with normal scores on PVF but deficits on category fluency, semantic verbal fluency qualify as differential deficits relative to semantic memory.
Although the lot of participants is not large, it brings important information about the particularities of the verbal fluency task in Romanian language on normal population and on the persons with depression. The importance of this study resides also in the comparation of two different PVF versions and also SVF. Normative data for verbal fluency task for Romanian population is a challenge for future research studies.
Limitations
The reduced number of participants for this study might be an inconvenient. Still results of this study should not be distorted by this, as long as other researches on verbal fluency used a limited number of participants. The lack of the valid norms for healthy Romanian population on verbal fluency task –was also a limitation. Yet, the study uses intergroup comparation so norms are not mandatory.
CONCLUSIONS
The Romanian sample of persons with depression showed deficits in cognitive functioning reflected by the total score of verbal fluency test, similar with others research.
In the case of person with depression it is necessary to apply both examination of verbal fluency (phonemic and semantic verbal fluency test). The results are valid indicators for cognitive functioning and can help in establishing a differential diagnosis.
Quantitative scoring of verbal fluency should be followed by qualitative scoring system.
ABBREVIATIONS
VFT – Verbal fluency task
PVF – Phonemic verbal fluency
SVF- Semantic verbal fluency
COWAT – Control oral word association test
FAS – Letters F, A, S – a version of Phonemic verbal fluency
CFL – Letters C, F, L – a version of Phonemic verbal fluency
CTL – Letters C, T, L – a version of Phonemic verbal fluency
PRW – CFL – Letters P, R, W – a version of Phonemic verbal fluency
MADRS – Montgomery & Asberg Depression Rating Scale
CVLT – California Verbal Learning Test
REFERENCES
1. Weiland-Fiedler P. Neuropsychological impairment in depression:
state or trait marker? Universitat Wien, 2005 (Internet) (cited 2015 Jul 13).
2. Fama R, Sullivan EV, Shear PK et al. Structural brain correlates of verbal and nonverbal fluency measures in Alzheimer’s disease. Neuropsychology 2000;14(1): 29–40.
3. Henry JD, Crawford JR, Phillips LH. Verbal fluency performance in dementia of the Alzheimer’s type: A meta-analysis. Neuropsychologia 2004;42(9): 1212–22.
4. Merens W, Booij L, Van Der Does AJW. Residual cognitive impairments in remitted depressed patients. Depress Anxiety 2008;25(6): E27–36. (cited 2015 Jul 13) Available from: http://www.ncbi.nlm.nih.gov/pubmed/18008314
5. Okada G, Okamoto Y, Yamashita H et al. Attenuated prefrontal activation during a verbal fluency task in remitted major depression: Short Communication. Psychiatry Clin Neurosci 2009;63(3): 423–5.
6. Hartinger B. Functional and structural imaging phenotypes of the amygdala and the cingulate cortex : an investigation in remitted depressed patients. Functional and structural, 2010.
7. Neumeister A, Konstantinidis A, Stastny J et al. Association between serotonin transporter gene promoter polymorphism (5HTTLPR) and behavioral responses to tryptophan depletion in healthy women with and without family history of depression. Arch Gen Psychiatry 2002;59(7):
6 1 3 – 2 0 . A v a i l a b l e f r o m :
http://www.ncbi.nlm.nih.gov/pubmed/12090814
8. Benton A. Multilingual aphasia examination manual of instructions,
1989.
9. Bowie CR, Harvey PD, Moriarty PJ et al. A comprehensive analysis of verbal fluency deficit in geriatric schizophrenia. Arch Clin Neuropsychol
2004;19(2): 289–303.
10. Araujo NB, Barca ML, Engedal K et al. Verbal fluency in Alzheimer’s disease, Parkinson’s disease, and major depression. Clinics (Sao Paulo)
2 0 1 1 ; 6 6 ( 4 ) : 6 2 3 – 7 . ( I n t e r n e t ) . A v a i l a b l e f r o m : http://www.pubmedcentral.nih.gov/articlerender .fcgi?artid=3093793& tool=pmcentrez&rendertype=abstract
11. Brébion G, Villalta-Gil V, Autonell J et al. Cognitive correlates of verbal memory and verbal fluency in schizophrenia, and differential effects of various clinical symptoms between male and female patients. Schizophr Res 2013;147(1): 81–5.
12. Castelli L, Lanotte M, Zibetti M et al. Apathy and verbal fluency in
STN-stimulated PD patients: An observational follow-up study. J Neurol
2007;254(9): 1238–43.
13. Henry JD, Beatty WW. Verbal fluency deficits in multiple sclerosis.
Neuropsychologia 2006;44(7): 1166–74.
14. Ho AK, Sahakian BJ, Robbins TW et al. Verbal fluency in Huntington’s disease: a longitudinal analysis of phonemic and semantic clustering and switching. Neuropsychologia 2002;40(8): 1277–84.
15. Baldo JV, Shimamura AP, Delis DC, Kramer J, Kaplan E. Verbal and design fluency in patients with frontal lobe lesions. J Int Neuropsychol Soc 2001;7(5): 586–96.
16. Henry JD, Crawford JR. A meta-analytic review of verbal fluency performance following focal cortical lesions.
Neuropsychology 2004;18(2): 284–95.
17. Henry JD, Crawford JR. A meta-analytic review of verbal fluency performance in patients with traumatic brain injury.
Neuropsychology 2004;18(4): 621–8.
18. Ruff RM, Evans R, Marshall LF. Impaired verbal and figural fluency after head injury. Arch Clin Neuropsychol 1986;1(2): 87–101.
19. Strauss E, Sherman EMS, Spreen O. A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary ( I n t e r n e t ) . 2 0 0 6 ( c i t e d 2 0 1 5 J u l 1 3 ) . Av a i l a b l e f r o m :
https://books.google.at/books/about/A_Compendium_of_Neuropsycho logical_Tests.html?id=jQ7n4QVw7-0C&pgis=1
20. Hurks PPM, Hendriksen JGM, Vles JSH et al. Verbal fluency over time as a measure of automatic and controlled processing in children with ADHD. Brain Cogn 2004;55(3): 535–44.
21. Loonstra AS, Tarlow AR, Sellers AH. COWAT metanorms across age, education, and gender. Appl Neuropsychol 2001;8(3): 161–6. (cited
2 0 1 5 J u l 1 3 ) A v a i l a b l e f r o m : http://www.ncbi.nlm.nih.gov/pubmed/11686651
22. Becker HE, Nieman DH, Dingemans PM et al.. Verbal fluency as a possible predictor for psychosis. Eur Psychiatry 2010;25(2): 105–10.
23. Norris MP, Blankenship-Reuter L, Snow-Turek AL, Finch J. Influence of depression on verbal fluency performance. Aging Neuropsychol Cogn 1995;2(3): 206–15. (cited 2015 Jul 13)Available f r o m : http://www.tandfonline.com/doi/abs/10.1080/13825589508256598#.V aO4KBuqqko
24. Henry J, Crawford JR. A meta-analytic review of verbal fluency deficits in depression. J Clin Exp Neuropsychol 2005;27(1): 78–101.
25. Murphy KJ, Rich JB, Troyer AK. Verbal fluency patterns in amnestic mild cognitive impairment are characteristic of Alzheimer’s type dementia. J Int Neuropsychol Soc 2006;12(4): 570–4.
26. Monsch AU, Bondi MW, Butters N et al. Comparisons of verbal fluency tasks in the detection of dementia of the Alzheimer type. Arch Neurol 1992;49(12): 1253–8.
27. Chertkow H, Bub D. Semantic memory loss in dementia of Alzheimer’s type. What do various measures measure? Brain 1990;113(2): 397–417. (cited 2015 Jul 14) Available from: http://www.ncbi.nlm.nih.gov/pubmed/2328410
28. Fossati P, Guillaume LB, Ergis A-M, Allilaire J-F. Qualitative analysis of verbal fluency in depression. Psychiatry Res 2003;117(1): 17–24.
29. Troyer AK, Moscovitch M, Winocur G, Alexander MP, Stuss D. Clustering and switching on verbal fluency: The effects of focal frontal- and temporal-lobe lesions. Neuropsychologia 1998;36(6): 499–504.
30. Troyer AK, Moscovitch M, Winocur G. Clustering and switching as two components of verbal fluency: evidence from younger and older healthy adults. Neuropsychology 1997;11(1): 138–46.
31. Ross TP, Calhoun E, Cox T, Wenner C, Kono W, Pleasant M. The reliability and validity of qualitative scores for the Controlled Oral Word Association Test. Arch Clin Neuropsychol 2007;22(4): 475–88. (cited 2 0 1 5 J u l 1 3 ) Av a i l a b l e f r o m :
h t t p : / / w w w. n c b i . n l m . n i h.gov/pubmed/17317094
32. Abwender DA, Swan JG, Bowerman JT, Connolly SW. Qualitative analysis of verbal fluency output: review and comparison of several scoring methods. Assessment 2001;8(3): 323–38.
33. Okruszek Ł, Rutkowska A, Wilińska J. Clustering and Switching Strategies During the Semantic Fluency Task in Men with Frontal Lobe Lesions and in Men with Schizophrenia. Psychol Lang Commun 2013;17(1): 93–100. (cited 2015 Jul 14) Available from: http://www.degruyter.com/view/j/plc.2013.17.issue-1/plc-2013- 0006/plc-2013-0006.xml
34. Okada G, Okamoto Y, Morinobu S, Yamawaki S, Yokota N. Attenuated left prefrontal activation during a verbal fluency task in patients with depression. Neuropsychobiology 2003;47(1): 21–6.
35. Videbech P, Ravnkilde B, Kristensen S, Egander A, Clemmensen K, Rasmussen NA, et al. The Danish PET/depression project: Poor verbal fluency performance despite normal prefrontal activation in patients w i t h m a j o r d e p r e s s i o n . P s y c h i a t r y R e s – N e u r o i m a g i n g .
2003;123(1):49–63.
36. Chávez-Oliveros M, Rodríguez-Agudelo Y, Acosta-Castillo I et al. Semantic verbal fluency in elderly mexican adults: Reference values. Neurologia 2015;30(4): 189–94. (cited 2015 Jul 14) Available from: http://apps.webofknowledge.com.web.bcucluj.ro:2048/full_record.do? product=UA&search_mode=GeneralSearch&qid=1&SID=N1DhtJg5l qVnpTpJd74&page=2&doc=54
37. Cavaco S, Gonçalves A, Pinto C et al. Semantic fluency and phonemic fluency: Regression-based norms for the portuguese population. Arch Clin Neuropsychol 2013;28(3): 262–71.
38. Schaller G. Verbal fluency Tests in der neuropsychologischen Demenzdiagnostik, 2007.
39. Florentina H. Specificul patologiei cognitive din perspectiva antropologica a pacientilor varstnici afectati de boli cardiovasculare. Universitatea de Medicina si Farmacie „Carol Davila”, 2007. (cited 2015 J u l 1 4 ) A v a i l a b l e f r o m : h t t p : / / w w w . b i b l i o g r a f i a . r o
/item.php?cu=2012052564106
40. Burtea V, Ifteni P, Vârgă O, Teodorescu A. Analiza fluentei verbale in episodul acut de schizofrenie. JMB 2013;(1): 54–8. (cited 2015 Jul 14) Av a i l a b l e f r o m : h t t p : / / w e b b u t . u n i t b v. r o / j m b / J M B 2 0 1 3 n r 1/02_05_original burtea.pdf
41. Popescu C, Tudose C, Niculăiţă A. Adaptarea româneasca a probei de Memorie episodicā cu coduri semantice Ri-48. Rom J Psychiatry 2015;17(1): 22–30.
42. Lovibond SH, Lovibond PF, Perţe A, Albu M, Copaciu A. DASS : manual pentru scalele de depresie, anxietate şi stres. Cluj-Napoca : A.S.C.R (Asociaţia de Ştiinţe Cognitive din România), 2011.
43. Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry 1979;134(4): 382–9.
44. Ross TP. The reliability of cluster and switch scores for the Controlled Oral Word Association Test. Arch Clin Neuropsychol 2003;18(2):
1 5 3 – 6 4 . ( c i t e d 2 0 1 5 J u l 1 4 ) A v a i l a b l e f r o m : http://www.ncbi.nlm.nih.gov/pubmed/14591467
45. Azuma T. Working memory and perseveration in verbal fluency. Neuropsychology 2004;18(1): 69–77.
46. Fossati P, Guillaume LB, Ergis AM, Allilaire JF. Qualitative analysis of verbal fluency in depression. Psychiatry Res 2003;117(1): 17–24.
47. Boone KB, Lesser IM, Miller BL et al. Cognitive functioning in older depressed outpatients: Relationship of presence and severity of depression to neuropsychological test scores. Neuropsychology 1995: 390–8.
48. Bolla KI, Lindgren KN, Bonaccorsy C, Bleecker ML. Predictors of verbal fluency (FAS) in the healthy elderly. J Clin Psychol 1990;46(5):
623–8.
49. Rodriguez-Aranda C, Martinussen M. Age-related differences in performance of phonemic verbal fluency measured by Controlled Oral Word Association Task (COWAT): a meta-analytic study. Dev Neuropsychol 2006;30(2):697–717. (cited 2015 Jul 14) Available from: http://www.ncbi.nlm.nih.gov/pubmed/16995832
50. Troyer AK. Normative data for clustering and switching on verbal fluency tasks. J Clin Exp Neuropsychol 2000;22(3): 370–8
***




