SOMATIC COMORBIDITIES IN UNIPOLAR DEPRESSION
Abstract
The somatic comorbidities in unipolar depression are a potential risk not only for the unfavorable course, but also increase the social and economic burden of this disorder A study conducted on a group of 5054 patients with depressive disorders that were hospitalized in the Neuropsychiatric Hospital of Craiova over a period of 5 years has shown a relatively high frequency of cardiovascular diseases and hyperglycemia, especially in women. The unfavorable course of the mental disorder was significantly associated with a higher frequency of somatic comorbidities, more hospitalizations and a longer duration of hospitalization. These results demonstrate the importance of the multidisciplinary approach for any comorbid association between mental and somatic disorders to improve the outcome of the medical treatment and to ensure a good recovery of the depressive episode.
Introduction
Epidemiological research has emphasized the fact that, in the case of unipolar depression, there is a high rate of comorbid associations with various medical conditions, which may significantly complicate both the disease prognosis and the psychopharmacological treatment, as well as the quality of the therapeutic response. The presence of somatic comorbidities is a major risk factor for the outcome of psychiatric disorders, with a 1.6-3 times higher mortality rate in this category of patients (1) that require a specific approach regarding the primary care, specialized care and treatment priorities.
The depressive disorder is an important part of the cardiovascular pathology, given the fact that it constitutes a risk factor in the initial stages of the disorder, and it can also lead to an unfavorable prognosis (2). The epidemiological data show a nearly 20% prevalence of depressive disorders in patients also suffering from cardiovascular diseases, with three times more frequent instances of psychiatric disorders in these patients than in general population (3). Depression can be generated by the side effects of cardiology medication (beta-blockers, thiazide diuretics, angiotensin-converting-enzyme inhibitors, antiadrenergic agents, some antiarrhythmic medicines, digital drugs) (4, 5).
Another frequent comorbidity in depression is hyperglycemia, people suffering from diabetes having a three times higher risk of developing depressive disorders, compared to the general population. Almost 20% of the diabetic patients (6), and particularly those with Type 2 diabetes, have severe depressive symptoms (7) that increase with 86% the costs of care in these patients compared to the care of the patients with diabetes only (8). The comorbidity of these two medical conditions may also appear as a side effect of the antidepressant or antipsychotic medication used to treat depression (9). Depressive disorders occur in 10-27% of the people having survived a cerebrovascular accident, with symptoms occurring for approximately one year. Another
15-40% of the patients show signs of depression during the first two months after the stroke (10). The stroke mortality rate was also 50% higher in the case of depressive patients (11). Depression also occurs in over 25% of the patients having been diagnosed with cancer. However, the depressive symptomatology is often mistaken for the side effects of the corticosteroid treatment or chemotherapy, or for the cancer symptoms which cause a loss of appetite and weight, insomnia and fatigue (12). This leads to a high rate of underdiagnoses in 40-90% of the patients (13).
Depression is also associated with hepatitis C. This association is a significant factor for the negative outcome of both conditions, when the effects of the immunosuppressant or antidepressant drugs play an important part in the entire process (14). Also, associated with depressive disorders, obesity is related to the hyperactivity of the hypothalamic–pituitary–adrenal axis, to high cortisol levels and to the side effects of some psychotropic substances (antidepressants, antipsychotics, mood stabilizers). Also, the overweight depressive patients have cognitive dysfunction that is correlated with structural abnormalities of the brain at the level of the white matter, hypothalamus, hippocampus, cingulate gyrus and basal ganglia (15).
The depressive disorder is merely one of the manifestations of the neurodegenerative diseases prodromal and it is also a risk factor for the increase of dysfunctionalities generated by this type of disorders. In Alzheimer’s disease, depression occurs along with the Mild Cognitive Impairment (MCI) syndrome, and it has a significant impact in its development to the stage of a neurodegenerative disease (16). It was revealed in 40% of the patients during the first three years of their disease. In
24% of the cases the cognitive deficit improved after this initial stage (17). Depression also occurs in the prodromal stage of Parkinson’s disease, in 20-40% of the patients (18). It is a risk factor for the cognitive deterioration which is specific to this neurodegenerative disease (19). The prodromal stages of multiple sclerosis are associated with depressive symptoms in up to 40% of cases (20). An important part in this comorbid association is that of the side effects of the immunosuppressants used to treat the disease, leading to a 54% incidence of depression in the patients diagnosed with neurodegenerative disease (21). In Huntington’s disease, depressive symptomatology occurs in 30-60% of cases, along with structural abnormalities of the brain at a mesencephalic level and a relatively high, 3-7% rate of suicidal behavior (22).
Depression is an important comorbid association for the HIV infection, as it amplifies the immune deficit generated by this viral affection and it occurs from the very beginning of the disease, along with the cognitive deficit (23). The depressive disorder also leads to a nearly
20% increase of the suicide risk, up to approximately 70% (24). Within the context of chronic pain and of HIV d i a g n o s i s , d e p r e s s i o n a l s o d i s p l a y s a n x i o u s manifestations, in over 70% of cases (25). The severity of the symptoms can also be aggravated by the side effects of the antiretroviral medication.
Materials and methods
We conducted a retrospective study by reviewing the observation sheets of 5054 patients diagnosed with Unipolar Depression, according to the ICD-10 criteria, and treated in the Neuropsychiatric Hospital of Craiova, between January 1, 2008 and December 31, 2012. The study complied with the ethical norms regarding clinical research and it was approved by the Local Ethics Committee.
Results
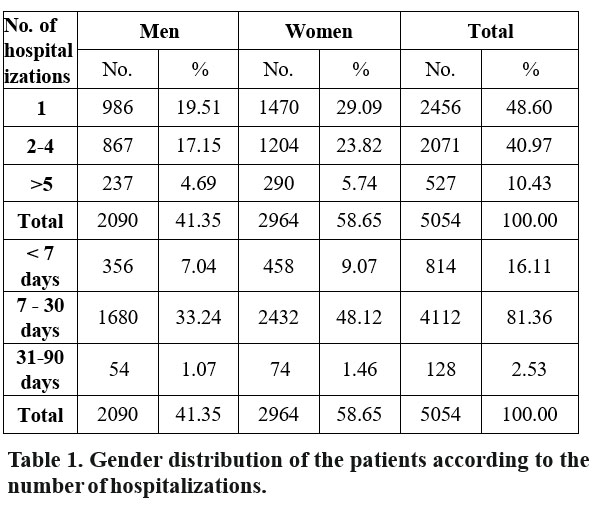
Between January 1, 2008 and December 31, 2012, there were 18306 hospitalizations in the two psychiatric clinics of the Neuropsychiatric Hospital in Craiova. 11168 (61.01%) of all admissions were treated for Unipolar Depression Disorder (UDD) with a total of 5054 patients that were included in our study. The distribution by gender showed a slightly higher proportion of women (2964;
58.65%) against men (2090; 41.35%). The average age was 52.37+6.28 years (53.94+5.82 years for men and 51.21+6.36 years for women) (p<0.001), and the predominant age group was 45-64 years with 1841 (36.43%) men and 2487 women (49.21%) respectively (Chi-square p < 0.001).
Almost half of the patients (2456; 48.60%) required only one hospitalization during the study period, while 527 patients (10.43%) were admitted more than 5 times for recurrent episodes of depression. Regarding the duration of the hospitalization, most of the patients (81.36%) ranged between 7 and 30 days, while a small proportion (2.53%) had longer hospitalizations (1-3 months) (Table 1).
Almost half of the patients included in the study group had at least one associated somatic disease. (2401 patients; 47.50%). Out of the total number of patients diagnosed with multiple disorders (both psychiatric and somatic), the majority had one (935 patients; 18.50%) or two somatic comorbidities (900 patients; 17.80%) (Table2).
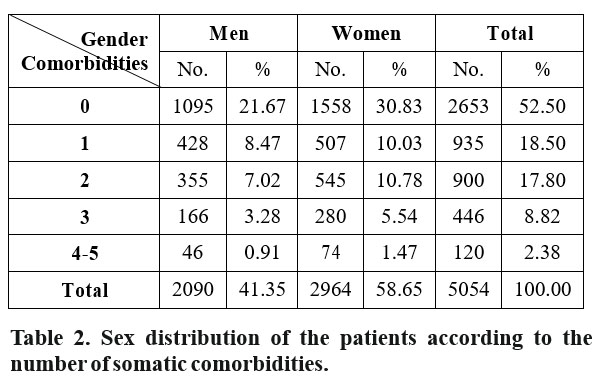
The comparative analysis of the number of comorbidities based on the gender of the patients showed a higher prevalence in women (58.65%, p<0.05).
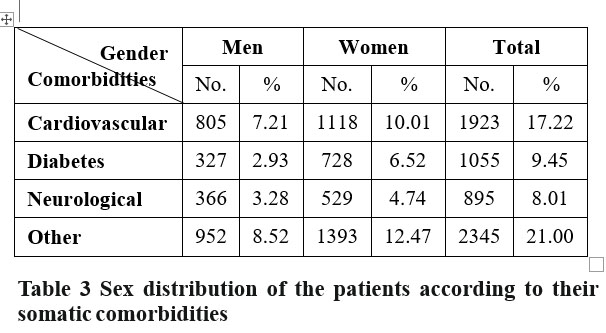
The cardiovascular comorbidities (myocardial infarction, arterial hypertension, coronary diseases, cardiac insufficiency) had the highest frequency (17.22%) with 10.01% women and 7.21% men. Over the 5 years under study, hyperglycemia was mentioned in 1055 instances (9.45%), with twice as many women (6.52%) as men (2.93%) (p<0.001), while other neurological and neurodegenerative disorders were recorded in 895 cases (8.01%), (4.74% women and 3.28% men, respectively).
The other somatic disorders (endocrine, metabolic, hepatic, infectious) found in the medical documentation, but not considered as significant were listed in the category of “other comorbidities” accounted for 2345 (21.00%) of the total number of hospitalizations included in the study. We noticed a slight tendency of associating unipolar depressive disorders with other comorbidities in women (p<0.05) (Table 3).
Discussions
The association between somatic comorbidities and major depressive disorder is a risk factor for the negative course of both nosologically entities, leading to higher mortality rates and to the need for complex care (1). Moreover, the comorbid association of two or more somatic disorders leads to an amplified debilitating effect of the psychiatric disease. This is reflected in the lower social and economic standard of the depressive patients, in the higher burden of disease and in the increase of care costs (26, 27).
The prevalence of comorbid cardiovascular diseases (arterial hypertension, myocardial infarction, cardiac insufficiency) found in our study is in accordance with data reported in the literature (3), while the prevalence of type I and type II diabetes (9.45%) and neurological complications (8.01%) are at the lower end of the published reports (6, 7, 10, 28).
The statistical analysis of the results emphasized the connection between the number of the somatic comorbidities associated with depressive disorders and the number of hospitalizations. Data suggest that a higher number of somatic comorbidities may complicate the course of the psychiatric disorder, which in turn increase the number and duration of admissions.
We found a significant correlation (p<0.001) between the number of comorbidities and number of hospitalizations (Figure 1).
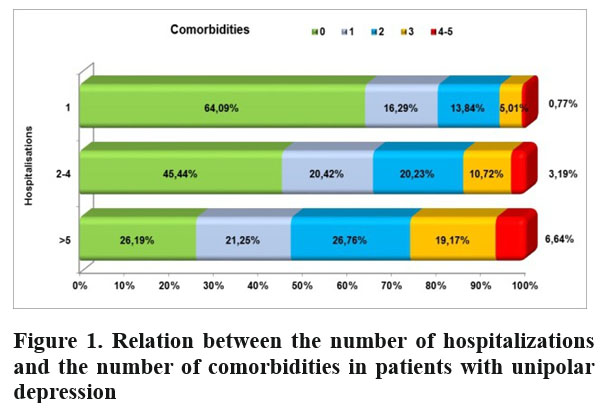
Thus, the proportion of patients requiring more than 5 hospitalizations compared to only one h o s p i t a l i z a t i o n i n c r e a s e s w i t h t h e n u m b e r o f comorbidities (26.76% vs. 13.84% for 2 comorbidities;
19.17% vs. 5.01% for 3 comorbidities; 6.64% vs. 0.77% for more than 3 comorbidities). The large number of admissions may also be related to the debilitating potential of depression (29), to higher care costs (27), as well as to a lower quality of life and to the perception of the disease as a burden (1, 30). The data suggest that the association of somatic comorbidities may aggravate the psychiatric condition, leading to more hospitalizations and to negative clinical, social and economic effects (31, 32).
The number of comorbidities associated with psychiatric disorders has also constituted one of the variables used to identify potentially significant d i ff e r e n c e s b e t w e e n t h e a v e r a g e d u r a t i o n o f hospitalization and the number of related somatic diseases. We searched for a set of statistically valid indicators for the influence of comorbidities on the course of the psychiatric disorder, which could predict an unfavorable outcome of the mental illness and a significant increase of the mortality rate (1).
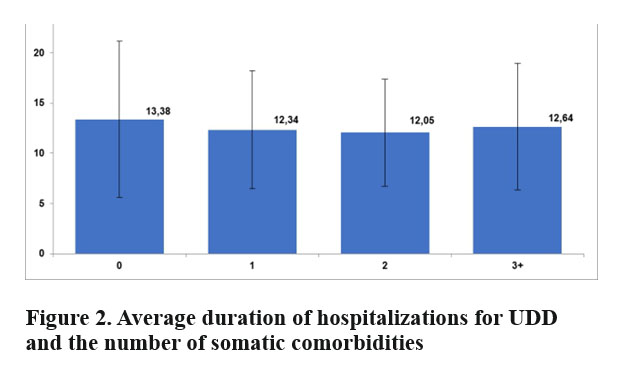
We found a longer average duration of admissions in patients with no somatic comorbidities (13.38+7.78 days), as compared to those with simultaneous manifestations of both the depressive and the somatic symptomatology (12.34+5.90 days for one somatic comorbidity, 12.05+5.35 days for two somatic comorbidities and 12.64+6.31 days in patients with three or more somatic comorbidities associated with the primary diagnosis of unipolar depression (p<0.001) (Figure 2).
Conclusions
Somatic comorbidities (mainly cardiovascular diseases and hyperglycemia) associated with major depression disorders occurred more frequently in the case of women (p<0.05). The number and average duration of hospitalizations, especially in the presence of somatic comorbidities, may be an indicator for the course of depression, with a significant correlation with the number and the duration of hospital admissions. (p<0.001). Within this context, it is imperative to approach this category of patients from a multidisciplinary angle, in order to obtain positive results from the therapeutic management process.
References
1.Harris EC, Barraclough B. Excess mortality of mental disorder. Br J Psychiatry 1998 Jul;173:11–53.
2.Ariyo AA, Haan M, Tangen CM, Rutledge JC, Cushman M, Dobs A, Furberg CD. Depressive symptoms and risks of coronary heart disease and mortality in elderly Americans: Cardiovascular Health Study Collaborative Research Group. Circulation 2000;102:1773–9
3.Joynt KE, Whellan DJ, O’Connor CM. Depression and cardiovascular disease: mechanisms of interaction. Biol Psychiatry 2003;54:248 – 61
4.Ko DT, Hebert PR, Coffey CS, Sedrakyan A, Curtis JP, Krumholz HM. Beta-Blocker Therapy and Symptoms of Depression, Fatigue, and Sexual Dysfunction, JAMA 2002; 288:351-357
5.Fourlanos S, Greenberg P. Managing drug-induced hyponatraemia in adults, Australian Prescriber, Vol. 26 No. 5 2003 6.Gavard JA, Lustman PJ, Clouse RE. Prevalence of depression in adults with diabetes. Diabetes Care 1993;16:1167-1178.
7.Leedom LLL, Meehan WP, Procci W, Zeidler A. Symptoms of depression in patients with type II diabetes mellitus. Psychosomatics 1991;32:280-286.
8.Ciechanowski PS, Katon WY, Russo JE. Depression and diabetes: Impact of depressive symptoms on adherence, function, and costs.
Arch Intern Med 2000;160:3278 3285.
9.McIntyre RS, Mancini DA, Pearce MM, Silverstone P, Chue P, Misener VL, Konarski JZ. Mood and Psychotic Disorders and Type 2
Diabetes: A Metabolic Triad, Canadian Journal of Diabetes 2005; 29(2):122-132
10.National Institute of Mental Health. Co-occurrence of Depression w i t h S t r o k e , A v a i l a b l e a t h t t p : / / www.nimh.nih.gov/depression/co_occur/stroke.htm. Consulted on July 12, 2014
11.Everson SA, Roberts RE, Goldberg DE, Kaplan GA: Depressive Symptoms and Increased Risk of Stroke Mortality Over a 29-Year Period, Archives of Internal Medicine 1998; 158:1133-1138.
12.National Institute of Mental Health. Co-occurrence of Depression w i t h C a n c e r , A v a i l a b l e a t h t t p : / / www.nimh.nih.gov/depression/co_occur/cancer.htm. Consulted on July 15, 2014
13.Singer S, Bringmann H, Hauss J, Kortmann RD, Köhler U, Krauss O, Schwarz R. Prevalence of concomitant psychiatric disorders and the desire for psychosocial help in patients with malignant tumors in an acute hospital, Dtsch Med Wochenschr 2007, 132:2071-6.
14.Lim JK, Cronkite R, Goldstein MK, Cheung RC. The Impact of Chronic Hepatitis C and Comorbid Psychiatric Illnesses on Health- related Quality of Life, J Clin Gastroenterol 2006;40:528–534.
15.Raji CA, Ho AJ, Parikshak NN, Becker JT, Lopez OL, Kuller LH, Hua X, Leow AD, Toga AW, Thompson PM. Brain Structure and Obesity, Hum Brain Mapp. 2010 Mar; 31(3): 353–364.
16.Andersen K, Lolk A, Kragh-Sorensen P, Petersen NE, Green A. Depression and the Risk of Alzheimer Disease, Epidemiology 2005;16: 233–238.
17.Holtzer R, Scarmeas N, Wegesin DJ, Albert M, Brandt J, Dubois B, Hadjigeorgiou GM, Stern Y. Depressive Symptoms in Alzheimer’s Disease: Natural Course and Temporal Relation to Function and Cognitive Status, J Am Geriatr Soc, 53:2083–2089, 2005.
18.Starkstein SE, Preziosi TJ, Bolduc PL, Robinson RG. Depression in Parkinson’s Disease. J Nerv Ment Dis. 1990; 178(1):27-31.
19.Aarsland D, Andersen K, Larsen JP, et al. Prevalence and characteristics of dementia in Parkinson disease: an 8-year prospective study. Arch Neurol. 2003; 60(3):387-392.
20.Sullivan MJ, Weinshenker B, Mikail S, Bishop SR. Screening for major depression in the early stages of multiple sclerosis. Can J Neurol Sci 22: 228-231.
21.Sadovnick AD, Remick RA, Allen J, Swartz E, Yee IM, Eisen K, Farquhar R, Hashimoto SA, Hooge J, Kastrukoff LF, Morrison W, Nelson J, Oger J, Paty DW. Depression and multiple sclerosis. Neurology, 1996, 46: 628-632.
22.Krogias C, Strassburger K, Eyding J, Gold R, Norra C, Juckel G, Saft C, Ninphius D. Depression in patients with Huntington disease correlates with alterations of the brain stem raphe depicted by transcranial sonography J Psychiatry Neurosci 2011;36(3):187-94.
23.Price RW, Spudich S. Antiretroviral therapy and central nervous system HIV type 1 infection. J Infect Dis 2008, 197(Suppl 3):S294-S306.
24.Robertson K, Parsons TD, Van Der Horst C, Hall C, Thoughts of D e a t h a n d S u i c i d a l I d e a t i o n i n N o n p s y c h i a t r i c H u m a n Immunodeficiency Virus Seropositive Individuals, Death Studies, 30: 455–469, 2006
25.Savetsky JB, Sullivan LM, Clarke J et al. Evolution of depressive symptoms in human immunodeficiency virus-infected patients entering primary care. Journal of Nervous and Mental Disease, 2001, 189, 76-83.
26.World Health Organization. The global burden of disease: 2004 u p d a t e . 2 0 0 4 A v a i l a b l e a t h t t p : / / www.who.int/entity/healthinfo/global_burden_disease/GBD_report_2 004update_full.pdf (Consulted on July 28, 2014)
27.Greenberg PE, Kessler RC, Birnbaum HG, et al. The economic burden of depression in the U.S.: how did it change between 1990 and 2000? J Clin Psychiatry 2003;64(12):1465–75.
28.Regenold WT, Thapar RK, Marano C, Gavirneni S, Kondapavuluru PV. Increased prevalence of type 2 diabetes mellitus among psychiatric inpatients with bipolar I affective and schizoaffective disorders independent of psychotropic drug use. J Affect Disord 2002; 70:19-26.
29.Murray CJL, Lopez AD, editors. The Global Burden of Disease: a comprehensive assessment of mortality and disability from diseases, injuries, and risk factors in 1990 and projected to 2020. Cambridge (Massachusetts): Harvard School of Public Health on behalf of the
World Health Organization & the World Bank, 1996.
30.Rotermann M. Marital breakdown and subsequent depression. Health Rep. 2007;18:33–44.
31.Kemp DE, Sylvia LG, Calabrese JR, Nierenberg AA, Thase ME, Reilly-Harrington NA, Ostacher MJ, Leon AC, Ketter TA, Friedman ES, Bowden CL, Rabideau DJ, Pencina M, Iosifescu DV; The LiTMUS Study Group. General medical burden in bipolar disorder: findings from the LiTMUS comparative effectiveness trial. Acta Psychiatr Scand 2014;129:24-34.
32.McIntyre RS, Konarski JZ, Soczynska JK, Wilkins K, Panjwani G, Bouffard B, Bottas A, Kennedy SH. Medical comorbidity in bipolar disorder: implications for functional outcomes and health service utilization. Psychiatr Serv 2006; 57:1140-1144.
***



