THE BEHAVIORAL APPROACH SYSTEM AS AN INSTRUMENT FOR ASSESSING CLINICAL STAGING IN PATIENTS WITH BIPOLAR I DISORDER
Abstract
Introduction: Bipolar disorder (BPD) is the sixth leading cause of disability worldwide. A staging model in BPD would be useful in order to provide the necessary information to the clinician for selecting stage-specific strategies for treatment. Multiple studies emphasize the view that the behavioral approach system (BAS) may be a relevant marker of illness onset and progression in BPD. Objective: The aim of this research study was to further explore the roles of self-reported BAS Drive, Fun Seeking and Reward Responsiveness levels in a sample of Romanian bipolar I disorder patients compared with a control group. Materials and methods: Our sample consisted of 93 subjects, divided in to 58 BPD I patients and 35 healthy controls. Consequently the BPD I group was subdivided, according to specific psychopathological clinical states such as mania, hypomania, depressive and mixed episodes, by using the Romanian version of the MINI International Neuropsychiatric Interview. The BIS/BAS self-report scale was used in order to assess the behavioural approach system. Results: Our study showed higher mean rank values of BAS score for the participants suffering from BPD I compared with healthy controls. Our data revealed increased mean rank values of BAS scores only for the participants who presented in a manic episode when compared with the depression, remission and the healthy control subgroups. High BAS Drive and BAS Reward Responsiveness scores were directly correlated with a greater number of past manic episodes, while a high BAS Fun Seeking score was indirectly correlated with the number of past episodes of depression. Conclusion: Our results contribute the current research findings regarding the BAS dysregulation theory in BPD and subscribe to the importance of measuring BAS hypersensitivity as a reliable behavioural marker for attaining a theoretically driven model for disease progression in BSD and improving therapeutic strategies for our patients.
INTRODUCTION
Approximately 4.4% of the population will experience some form of bipolar spectrum disorder (BSD) in their lifetime (1). The lifetime prevalence of BPD I is approximately 1.0% and, respectively 1.1% for BPD II (1). However, sub-threshold screening probably escalates the burden of disease in the general population (2). Patients suffering from BPD have an increased risk for suicide, substance abuse and severe functional impairment (3). Considering that BPD is the sixth leading cause of disability worldwide one must also factor in the significant cost its insufficient management may infer on society(4).
Bipolar disorder is a heterogeneous condition, characterized by symptoms that span from mild cyclothymia to severe depression or full-blown mania, sometimes accompanied by psychotic features (5-7). Due to the wide variety of illness presentation and progression of the bipolar spectrum disorders, clinical staging, alongside conventional diagnostic models, greatly contributes to the prognostic validity in BPD (8). A staging model in BPD would be useful in order to specify where an individual is positioned on the continuum from ‘at risk’ but asymptomatic to ‘end-stage’ illness and to provide the necessary information to the clinician for selecting stage- specific strategies for treatment (8, 9). Research data has revealed multidimensional interactions in BPD that
involve genetic, physiological, emotional, cognitive, behavioral and environmental factors (10-12). A viable model for clinical staging in bipolar disorder needs to be able to take into account a multisystem perspective of the disease progression and incorporate multiple domains, such as biological, cognitive, behavioral, in order to fully provide a prognostic validity (8). The allostatic load framework provides a suitable foundation for a clinical staging model in bipolar disorder, by integrating the exogenous stressors with physiological functioning (8, 13). The notion of ‘allostatic load’ refers to the cumulative deterioration of the organisms’ capacity to adjust in anticipation of and in response to various stressors (13, 14). Several research groups propose the dysregulation of the behavioral approach system (BAS) as a relevant marker for assessing the allostatic load of disease progression in BPD (8, 10). The behavioral approach system, also known as the reward system, was first described as part of a three-system model of emotion, motivation and learning, together with the behavioral inhibition system (BIS) and the fight/flight system (FFS) (15). According to Gray, it is the activity of these three behavioral state systems that produce longer-term personality traits and predispose individuals to respond in a predictable manner to different circumstances (15). BAS is postulated to regulate approach motivation and goal- directed behavior to attain rewards, and is associated with positive affect, hope and anticipation of pleasure (16), as well as with anger, when the goal pursuit proves to be unattainable (17, 18). The associated personality dimensions of BAS are optimism, reward-orientation, impulsiveness, while the pathological manifestations of this system may result in addictive behaviors, impulsive behaviors or mania(19-21).
The relevance of BAS in BPD originated, on one hand, from the core symptoms of mania and depression, expressed by locomotor and incentive-reward, and on the other, from the neurobiological underpinnings of BAS to the dopaminergic projections from the A10 nucleus in the ventral tegmental area (VTA) to the frontal cortex, amygdala, nucleus accumbens, ventral pallidum, septum and hippocampus (22, 23). The early formulation of the BAS dysregulation theory of bipolar spectrum disorders (BSD) hypothesize that hypomanic/manic and depressive states are opposite reflections of an overactive BAS for the former and respectively, a decreased activation of BAS for the later (22). Moreover, it is posited that a hyperactive BAS is a trait-like feature, and individuals suffering from BSD may have a genetically predetermined mean level of BAS that predicts the kind of progression the disease may have towards manic or depressive states (22, 24). More recent studies add to this research by suggesting that relevant environmental stimuli, involving rewards or goal seeking and attainment, underlined by a hyperactive BAS, lead to excessive reward motivation and approach-related affect, which in turn lead to hypomanic or manic symptoms(25, 26). Likewise, BDS individuals experience an excessive decrease in BAS, in response to non- attainment of goals or rewards, which leads to depressive symptoms (25, 26). Furthermore, it is postulated that people suffering from BSD not only react more strongly to significant life events but also their BAS hypersensitivity steers them towards goal-seeking and reward attaining situations which, in turn, increase their chances of developing an affective episode(27-29).
Several clinical instruments of BAS sensitivity have been developed to test the BAS dysregulation theory. Such a measure is the BIS/BAS self-report scale by Carver and White, which is comprised of three BAS components. The first, measures the persistent pursuit of desired goals (BAS Drive), the second, assesses the desire for new rewards and a willingness to approach potentially rewarding events (Fun Seeking), while the third focuses on positive responses to the occurrence or in anticipation of reward (Reward Responsiveness).(30)
In order to better advance current scientific knowledge of BPD and attain a theoretically-driven model for disease progression, we need to further investigate bio- behavioral markers and test for evidence based approaches. This, in turn, will yield better prognostic value and improve therapeutic strategies for our patients.
The aim of this research study was to further explore the roles of self-reported BAS Drive, Fun Seeking and Reward Responsiveness levels in a sample of Romanian bipolar I disorder patients compared to a control group.
MATERIAL AND METHOD
Participants
The patients included in this research fulfilled the Diagnostic and Statistical Manual of Mental Disorders IV Text Revision (DSM-IV-TR) (31), diagnostic criteria for BPD I. The diagnostic was confirmed by administering the Romanian version of the MINI International Neuropsychiatric Interview (M.I.N.I. 6.0)(32) by a trained psychiatrist, for each participant. The M.I.N.I. was also used in order to classify the BPD I sample according to specific psychopathological clinical states such as mania, hypomania, depressive and mixed episodes. Organic mental disorders, mental retardation, schizophrenia or other psychotic disorders, other affective disorders, uncontrolled or serious medical conditions, pregnancy or postpartum period were used as exclusion criteria. The study group, comprised of 75 subjects, aged between 25 and 65, was recruited from the ‘Prof. Dr. Al. Obregia’ Clinical Psychiatric Hospital, during an eight-month period.
The control group was comprised of 35 participants and was selected from individuals that attended the outpatient clinic of ‘Prof. Dr. Al. Obregia’ Clinical Psychiatric Hospital to be certified as mentally healthy by a psychiatrist, received this certification, which was reconfirmed by M.I.N.I upon entering the research.
Informed consent was obtained from each participant, after a detailed description of the study. Clinical assessments were performed just once, for each subject, during a three-day interval.
Clinical assessments
A socio-demographical and clinical data semi- structured questionnaire, developed and used specifically for this research, was administered. It recorded specific characteristics of the sample: type of presentation (in- patient/out-patient), number of voluntary/involuntary past admissions, present clinical state, medical history, substance use, presence of past legal issues, age of onset for BPD I disorder, number and types of past episodes, stressful life events, family history of suicide or psychiatric disorders and current pharmacological treatment. Assessment of suicide thoughts and behaviours was performed using the Romanian version of Columbia- Suicide Severity Rating Scale (C-SSRS, Baseline Version 1/14/09)(33).
The clinical assessment of the behavioural approach system (BAS) was performed using the BAS items of the BIS/BAS self-report scale (30). This semi-structured questionnaire consists of 24 questions measured on a 4 point Likert scale, ranging from 1 = ‘ very true for me’ to 4 = ‘very false for me’. Four items are fillers, 7 questions assess the behavioural inhibition system (BIS) and the remaining 13 assess three of the BAS components as follows: 4 items for BAS Drive, 4 items for BAS Fun Seeking (BAS_FS) and 5 items for BAS Reward Responsiveness (BAS_RR).
Statistical analysis
For statistical analysis we used the IBM SPSS v22 statistical package. Data was tested for normality using the Shapiro-Wilk test and by assessing Q-Q plots and histogram data distribution. For our analysis, participants were grouped according to the Romanian version of the M.I.N.I. 6.0 criteria into controls and subjects suffering from BPD I. The BPD I sample was subsequently divided according to the affective state during presentation as follows: ‘mania’, ‘hypomania’, ‘depression’, ‘remission’ and ‘mixed’ subgroups.
In order to test for differences in BAS scores across our groups we performed independent samples t- student and Mann-Whitney U tests. Statistical significance was set at p < .05 with a confidence interval (CI) of 95%.
RESULTS
Demographical and clinical characteristics
The sample consisted of 75 patients diagnosed with BPD I and 35 controls (N = 110). Thirteen participants were removed because of insufficient data for the BAS scores (N = 97). The mean age in our total sample was 39.30 years (SD = 12.308) and there was a higher prevalence of females (N = 65, 67%) over males (N = 32,
33%). The study sample was comprised of 36.08% (N = 35) controls, 13.4% of the participants were in remission (N = 13), while the others were distributed according to current clinical episode as follows: 33% mania episode (N
= 32), 11.3% depressive episode (N = 14), 2.06% hypomanic episode (N = 2), and 4.1% with mixed episode (N = 4). Due to the low number of participants undergoing hypomanic episodes in our sample, we collapsed this category with mania. Moreover, we decided not to present the data regarding participants who experienced mixed episodes due to the paucity of cases and thus irrelevant statistical results. Thus, our final sample consisted of 93 subjects (N=93), divided in to 58 BPD I patients and 35 controls.
BAS scores in respect to different groups in our sample:
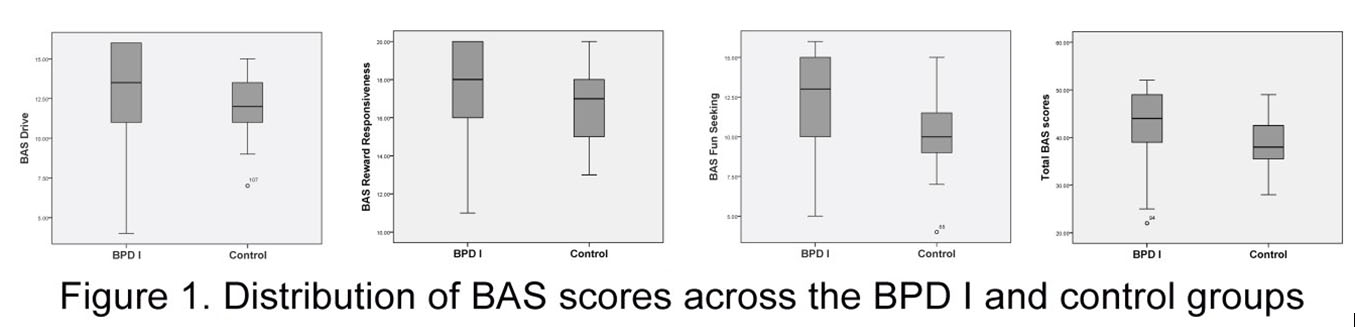
In our sample, patients suffering from BPD I had significant higher mean rank values of total BAS scores compared with the control group (N = 93, MR =
53.86 vs. 35.63, p = 0.002). Likewise, when analyzing the three subcomponents of the BAS, between the two groups, we found that the BPD I group displayed significant higher mean rank values of BAS Drive (N = 93, MR = 52.41 vs. 38.04, p= 0.012), BAS Reward
Responsiveness (BAS_RR) (N = 93, MR = 52.66 vs. 37.61, p = 0.008) and BAS Fun seeking (BAS_FS) (N = 93, MR = 54.09 vs. 35.24, p = 0.001) compared to the control group.
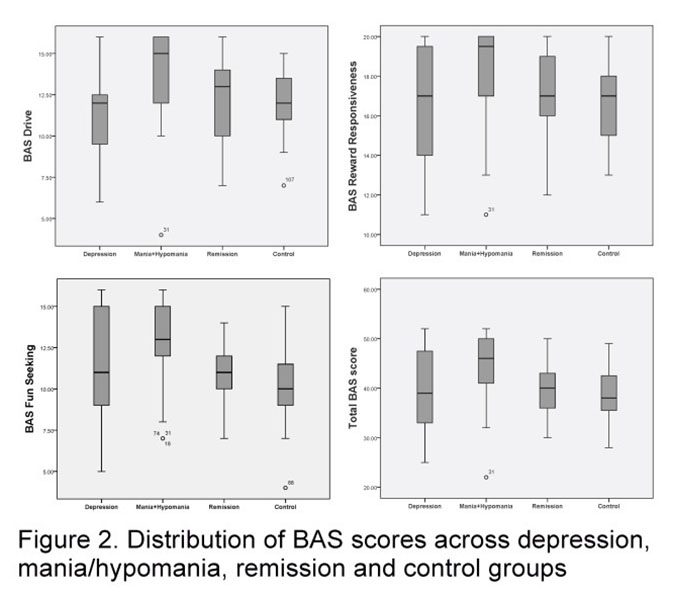
Furthermore, we analyzed the BAS scores across controls and the BPD I subgroups, distributed according to the current clinical state during presentation: ‘remission’,
‘depression’ and ‘mania/hypomania’. What we found was that individuals who presented with manic/hypomanic episodes have higher mean rank values of all BAS subcomponents scores when compared to controls (N =
67, (BAS_Drive_MR = 43.62 vs. 25.20, BAS_Drive_p =
0.0001); (BAS_RR_MR = 42.06 vs. 26.63, BAS_RR_p =
0.001); (BAS_FS_MR = 44.41 vs. 24.49, BAS_FS_p =
0.0001)). Likewise, we found significantly higher mean rank values of the BAS Drive and Bas Fun Seeking subcomponents in the ‘mania/hypomania’ subgroup when compared with the ‘remission’ subgroup (N = 45, (BAS_Drive_MR = 25.91 vs. 15.85, BAS_Drive_p =
0.018); (BAS_FS_MR = 26.45 vs. 14.50, BAS_FS_p =
0.005)), as well as higher BAS Drive mean rank values, when comparing ‘mania/hypomania’ with the ‘depression’ subgroup. (N = 43, BAS_Drive_MR = 24.53 vs. 14.64, BAS_Drive_p = 0.023). Our study didn’t yield any significant increase in BAS subcomponents’ scores, neither when comparing controls to the ‘remission’ or ‘depression’ subgroups, nor when comparing BAS values between the ‘remission’ and the ‘depression’ subgroups.
Further analysis of our sample, from a dynamic perspective, showed significantly increased BAS Drive (p = 0.027, r = 0.281) and BAS RR (p = 0.005, r = 0.349) scores for individuals who experienced a greater number of past manic episodes, while an increased BAS Fun Seeking score (p = 0.035, r = 0.269) was correlated with a smaller number of past depressive episodes.
DISCUSSIONS
In the present research study we found significantly higher mean rank values of BAS scores for the participants suffering from BPD I compared with healthy controls. Other research studies have also reported similar findings of greater BAS sensitivity in individuals with bipolar spectrum disorder compared with controls
(34). However, our results should be interpreted cautiously, considering the greater context. Firstly, one should keep in mind the fact that the ‘mania/hypomania’ subgroup was the best represented in our sample and thus might yield influence on the results. Secondly, when taking into account different clinical subgroups according to the affective state upon admission in the study and comparing them to the healthy control group, our data revealed increased mean rank values of BAS scores only for the ‘mania/hypomania’ subgroup. Likewise, the BAS mean ranks values were significantly higher solely for the
‘ m a n i a / h y p o m a n i a ‘ s u b g r o u p , w h e n c o m p a r i n g individuals in remission with those experiencing manic or depressive episodes, as well as when comparing ‘mania/hypomania’ with the ‘depression’ subgroup. From the clinical standpoint, the association between the mania episode and high self-rated BAS scores is not of great importance for BPD I staging purposes. Our study did not show any significant increase in BAS subcomponent scores neither when comparing controls to the ‘remission’ or ‘depression’ subgroups, nor when comparing between the ‘remission’ or ‘depression’ subgroups, thus not supporting the hypotheses that the higher BAS sensitivity may be independent of mood-related state episodes of illness (10). However, research studies that clearly establish an association between BSD and BAS hypersensitivity, either include larger samples of euthymic patients, or involve longitudinal designs (35- 37), both of which are not available in our research.
When examining scientific literature for data supporting self-reported reward sensitivity as a predictor for the course of BSD, results render conflicting findings. Consistent with the BAS dysregulation theory, some longitudinal studies, controlled for hypomanic/manic and depressive episodes, report that a higher BAS sensitivity scores anticipates a shorter time to relapse into a hypomanic/manic episode, while a higher BAS Reward Responsiveness marginally predicts a shorter relapse time of a depressive episode (35). Other studies rendered similar results regarding relapse of hypomanic/manic episodes following a higher baseline BAS sensitivity score, but reported lower BAS sensitivity values as a predictor for depressive episodes, when compared with asymptomatic patients (37, 38). The data that correlates future depressive episodes with a lower BAS sensitivity score is somewhat contradictory to the current BAS dysregulation theory. In order to provide a dimensional view upon the BAS values in our BPD I sample, we analysed the BAS Drive, BAS Reward Responsiveness and BAS Fun Seeking scores in respect to the number of past episodes of mania and depression. Our results showed increased BAS Drive and BAS Reward Responsiveness scores for individuals who experienced a greater number of past manic episodes, while an increased BAS Fun Seeking score was significantly correlated with a smaller past number of depressive episodes.
Nevertheless, several limitations of the current study need to be considered. First, clinical assessments were performed just once, thus more longitudinal data is needed in order to conclude on BAS hypersensitivity as a being associated or as being a predictor for BDS in our research. Second, our remission (euthymic) sample size was too small in order to properly control for specific BPD I psychopathological episodes.
In conclusion, the results of the present study have shown significant higher mean rank values of BAS scores for the individuals suffering from BPD I compared with healthy controls. Our data revealed increased mean rank values of BAS scores only for the participants who presented in a manic episode when compared with depression, remission and the healthy control subgroups. High BAS Drive and BAS Reward Responsiveness scores were directly correlated with a greater number of past manic episodes, while a high BAS Fun Seeking score was indirectly correlated with the number of past episodes of depression. Our results contribute the current research findings regarding the BAS dysregulation theory in BPD and subscribe to the importance of measuring BAS hypersensitivity as a reliable behavioural marker for attaining a theoretically-driven model for disease progression in BSD and improving therapeutic strategies for our patients.
Ethical Approval:
The Ethical Committee of ‘Prof. Dr. Al. Obregia’ Clinical Psychiatric Hospital in Bucharest, Romania, approved this research study.
Acknowledgements:
Authors would like to thank Andrei Crasan for his generous feedback, while writing the article.
Authors would like to respectfully thank fellow doctors from the ‘Prof. Al. Obregia Clinical Hospital’ for their support in recruiting the sample and thank all participants for their cooperation and sample contributions.
Abbreviations:
BSD – Bipolar spectrum disorder
BPD – Bipolar disorder
BAS – Behavioral approach system BIS – Behavioral inhibition system FFS – Fight/Flight system
BPD – Bipolar Disorder
VTA – Ventral tegmental area
BAS-FS – Behavioral approach system – Fun Seeking BAS-RR – Behavioral approach system – Reward Responsiveness
DSM IV-TR – Diagnostic and Statistical Manual of
Mental Disorders IV Text Revision
M.I.N.I – MINI International Neuropsychiatric Interview
C-SSRS – Columbia Suicide Severity Rating Scale
CI – confidence interval
N – Number of cases
REFERENCES
1.Merikangas KR, Akiskal HS, Angst J, Greenberg PE, Hirschfeld RM, Petukhova M, et al. Lifetime and 12-month prevalence of bipolar spectrum disorder in the National Comorbidity Survey replication. Arch Gen Psychiatry. 2007;64(5):543-52.
2.Glick ID. Undiagnosed Bipolar Disorder: New Syndromes and New Treatments. Primary Care Companion to The Journal of Clinical Psychiatry. 2004;6(1):27-33.
3.Murray CJ, Lopez AD. Evidence-based health policy–lessons from the Global Burden of Disease Study. Science. 1996;274(5288):740.
4.Kleinman L, Lowin A, Flood E, Gandhi G, Edgell E, Revicki D. Costs of bipolar disorder. Pharmacoeconomics. 2003;21(9):601-22.
5.Akiskal HS. The prevalent clinical spectrum of bipolar disorders:
beyond DSM-IV. Journal of clinical psychopharmacology. 1996;16(2):4S-14S.
6.Cassano GB, Dell’Osso L, Frank E, Miniati M, Fagiolini A, Shear K, et al. The bipolar spectrum: a clinical reality in search of diagnostic criteria and an assessment methodology. Journal of Affective disorders. 1999;54(3):319-28.
7.Shen GH, Alloy LB, Abramson LY, Sylvia LG. Social rhythm regularity and the onset of affective episodes in bipolar spectrum individuals. Bipolar disorders. 2008;10(4):520-9.
8.Kupfer DJ, Frank E, Ritchey FC. Staging bipolar disorder: what data and what models are needed? Lancet Psychiatry. 2015;2(6):564-70.
9.Kapczinski F, Magalhaes PV, Balanza-Martinez V, Dias VV, Frangou S, Gama CS, et al. Staging systems in bipolar disorder: an International Society for Bipolar Disorders Task Force Report. Acta Psychiatr Scand. 2014;130(5):354-63.
10.Alloy LB, Nusslock R, Boland EM. The development and course of bipolar spectrum disorders: an integrated reward and circadian rhythm dysregulation model. Annu Rev Clin Psychol. 2015;11:213-50.
11.McGuffin P, Rijsdijk F, Andrew M, Sham P, Katz R, Cardno A. The heritability of bipolar affective disorder and the genetic relationship to unipolar depression. Archives of general psychiatry. 2003;60(5):497- 502.
12.Johnson SL, Kizer A. Bipolar and unipolar depression: A comparison of clinical phenomenology and psychosocial predictors. 2002.
13.Bizik G, Picard M, Nijjar R, Tourjman V, McEwen BS, Lupien SJ, et al. Allostatic load as a tool for monitoring physiological dysregulations and comorbidities in patients with severe mental illnesses. Harv Rev Psychiatry. 2013;21(6):296-313.
14.McEwen BS, Stellar E. Stress and the individual. Mechanisms leading to disease. Arch Intern Med. 1993;153(18):2093-101.
15.Gray JA, McNaughton N. The neuropsychology of anxiety: An enquiry into the function of the septo-hippocampal system: Oxford university press; 2003.
16.McNaughton N, Gray JA. Anxiolytic action on the behavioural inhibition system implies multiple types of arousal contribute to anxiety. J Affect Disord. 2000;61(3):161-76.
17.Carver CS. Negative affects deriving from the behavioral approach system. Emotion. 2004;4(1):3-22.
18.Harmon-Jones E, Sigelman J. State anger and prefrontal brain activity: evidence that insult-related relative left-prefrontal activation is associated with experienced anger and aggression. J Pers Soc Psychol. 2001;80(5):797-803.
19.McNaughton N, Corr PJ. A two-dimensional neuropsychology of defense: fear/anxiety and defensive distance. Neurosci Biobehav Rev. 2004;28(3):285-305.
20.McNaughton N, Corr PJ. The neuropsychology of fear and anxiety: A foundation for reinforcement sensitivity theory. The reinforcement sensitivity theory of personality. 2008:44-94.
21.McNaughton N, Corr PJ. Animal cognition and human personality. The reinforcement sensitivity theory of personality Cambridge University Press,
Cambridge. 2008.
22.Depue RA, Iacono WG. Neurobehavioral aspects of affective disorders. Annu Rev Psychol. 1989;40:457-92.
23.Depue RA, Collins PF. Neurobiology of the structure of personality: Dopamine, facilitation of incentive motivation, and extraversion. Behavioral and Brain Sciences. 1999;22(03):491-517.
24.Depue RA, Krauss SP, Spoont MR. A two-dimensional threshold model of seasonal bipolar affective disorder. 1987.
25.Alloy LB, Abramson LY. The Role of the Behavioral Approach System (BAS) in Bipolar Spectrum Disorders. Curr Dir Psychol Sci. 2010;19(3):189-94.
26.Johnson SL, Edge MD, Holmes MK, Carver CS. The behavioral activation system and mania. Annu Rev Clin Psychol. 2012;8:243-67.
27.Hammen C. Generation of stress in the course of unipolar depression. J Abnorm Psychol. 1991;100(4):555-61.
28.Alloy LB, Abramson LY, Urosevic S, Bender RE, Wagner CA. Longitudinal Predictors of Bipolar Spectrum Disorders: A Behavioral Approach System (BAS) Perspective. Clin Psychol (New York). 2009;16(2):206-26.
29.Urošević S, Abramson LY, Harmon-Jones E, Alloy LB. Dysregulation of the behavioral approach system (BAS) in bipolar spectrum disorders: review of theory and evidence. Clinical psychology review. 2008;28(7):1188-205.
30.Carver CS, White TL. Behavioral inhibition, behavioral activation, and affective responses to impending reward and punishment: the BIS/BAS scales. Journal of personality and social psychology. 1994;67(2):319.
31.Association AP. Diagnostic and statistical manual of mental disorders: DSM-IV-TR. Washinton2000.
32.Sheehan D, Lecrubier Y, Sheehan KH, Janavs J, Weiller E, Keskiner A, et al. The validity of the Mini International Neuropsychiatric Interview (MINI) according to the SCID-P and its reliability. European Psychiatry. 1997;12(5):232-41.
33.Posner K, Brent D, Lucas C, Gould M, Stanley B, Brown G, et al. Columbia-suicide severity rating scale (C-SSRS). New York, NY: Columbia University Medical Center. 2008.
34.Urosevic S, Abramson L, Harmon-Jones E, Donovan P, Van Voorhis L, Hogan M, et al. The Behavioral Approach System (BAS) and bipolar spectrum disorders: Relationship of BAS and Behavioral Inhibition System (BIS) sensitivities to bipolar spectrum diagnoses and hypomanic personality. Manuscript under editorial review. 2006.
35.Alloy LB, Abramson LY, Walshaw PD, Cogswell A, Grandin LD, Hughes ME, et al. Behavioral approach system and behavioral inhibition system sensitivities and bipolar spectrum disorders: Prospective prediction of bipolar mood episodes. Bipolar disorders. 2008;10(2):310- 22.
36.Meyer B, Johnson SL, Winters R. Responsiveness to threat and incentive in bipolar disorder: Relations of the BIS/BAS scales with symptoms. Journal of Psychopathology and Behavioral Assessment. 2001;23(3):133-43.
37.Salavert J, Caseras X, Torrubia R, Furest S, Arranz B, Duenas R, et al. The functioning of the Behavioral Activation and Inhibition Systems in bipolar I euthymic patients and its influence in subsequent episodes over an eighteen-month period. Personality and Individual Differences.
2007;42(7):1323-31.
38.Hayden EP, Bodkins M, Brenner C, Shekhar A, Nurnberger Jr JI, O’Donnell B, et al. A multimethod investigation of the behavioral activation system in bipolar disorder. Journal of Abnormal Psychology. 2008;117(1):164.
***



